A single customer complaint about a hygiene product escalates. Soon, a potential defect is identified, but its origin is unknown. The question arises: do you recall a week’s worth of product, or an entire year’s? The financial and reputational difference is immense.
This scenario highlights the razor-thin margin for error in the North American B2B market. Here, ‘strict liability’ laws mean your company can be held responsible for a defective product even if you weren’t negligent. For procurement managers and compliance officers sourcing hygiene products, this legal reality places immense pressure on supplier selection and oversight.
This guide provides a practical framework for shielding your brand, detailing how to leverage supplier quality systems, full traceability, and third-party validation to build a defensible and resilient supply chain.
Your Liability Shield at a Glance
In North America’s strict liability environment, comprehensive documentation is not optional—it is your primary legal defense against potentially catastrophic claims.
Proactively vetting a supplier’s quality management system and demanding full batch traceability are the most effective actions you can take to mitigate risk.
Understanding the Stakes: Product Liability in the North American Market
The Principle of Strict Liability: Why ‘Fault’ Doesn’t Matter
Strict liability is a legal doctrine that shifts the focus from negligence to the mere fact that a product was defective and caused harm. In this context, a plaintiff does not need to prove that the manufacturer or supplier acted carelessly or failed to meet a standard of care; it is enough to show that the product was unsafe and directly led to injury or damages. This means that even if a hygiene product manufacturer has taken all reasonable precautions, they can still be held liable if a defect exists in the product.
Unlike negligence-based systems, where the injured party must demonstrate a breach of duty, strict liability imposes a higher risk on suppliers and manufacturers throughout the entire supply chain—including raw material suppliers, manufacturers, distributors, and retailers. In North America, this legal framework increases the importance of meticulous quality control and traceability because every entity involved can face liability regardless of fault, making a proactive and documented approach to product safety essential for risk mitigation.
Beyond the Courtroom: The Full Spectrum of Risk
Financial risks in product liability extend far beyond the initial claim. Legal defense costs alone can escalate quickly, often running into hundreds of thousands of dollars, even if the case is ultimately dismissed. Settlements and judgments can further drain company resources, and the cost of executing a product recall—including logistics, disposal, and lost inventory—can be devastating. For hygiene product manufacturers, these financial burdens can compromise not only short-term cash flow but also long-term viability.
Reputational risks pose equally significant challenges. A single high-profile incident can quickly erode customer trust, invite negative media scrutiny, and reduce brand equity built over years or decades. Damaged reputation often results in lost sales, decreased market share, and increased difficulty launching new products. Regulatory risks compound these pressures, as agencies like the U.S. Consumer Product Safety Commission (CPSC) or Health Canada can impose fines, sanctions, or mandatory corrective actions. These enforcement measures can disrupt operations and further harm public perception. Proactively managing these interconnected risks through effective enterprise risk management is essential to safeguard your company’s future.
Sustainable Hygiene Solutions for Your Business Needs
Top Source Hygiene offers eco-friendly and customizable hygiene products specifically designed for North American markets. From toilet paper to facial tissues, our solutions address sustainability, quality, and cost-effectiveness, making us your trusted partner in the industry.
Contractual Safeguards: The Role of the UCC
The Uniform Commercial Code (UCC) serves as the foundational legal framework governing commercial transactions across the United States, including sales of goods like hygiene products. It standardizes contract law to provide consistency and predictability in business dealings, making it essential for procurement and compliance officers to understand when negotiating agreements with suppliers. Clear contractual terms that specify warranties, indemnification obligations, and detailed product specifications are a critical first line of defense in allocating risk and defining each party’s responsibilities.
One particularly important aspect under the UCC is Section 2-207, often called the ‘Battle of the Forms.’ This provision addresses scenarios where the buyer and supplier exchange forms with conflicting or additional contract terms, which is common in supplier agreements. Without precise and mutually agreed-upon terms, such discrepancies can create legal uncertainties and disputes over liability. Therefore, rigorous contract review and negotiation to clarify all terms upfront help prevent costly misunderstandings and reinforce your company’s protection against product liability risks.
An Investment in Resilience, Not a Cost
While implementing a comprehensive quality and traceability system requires a proactive investment, it is best understood not as an operational cost, but as an investment in resilience. Consider the alternative: the cost of a single product recall can easily run into the millions of dollars when factoring in logistics, legal fees, regulatory fines, and lost sales—to say nothing of the long-term damage to brand reputation.
Viewed through this lens, the upfront expense of partnering with a supplier who has these systems embedded in their operations is a calculated, strategic expenditure. It is designed to safeguard your brand’s long-term value and market position, transforming a potential liability into a predictable and manageable part of your supply chain strategy.
Your First Line of Defense: The Quality Management System (QMS)
A supplier’s Quality Management System (QMS) is the foundational framework that ensures product safety and compliance by embedding quality into every step of the manufacturing process, rather than relying solely on final inspections. This structured system documents the processes, responsibilities, and controls that guarantee consistent adherence to quality standards, minimizing the risk of defects that could lead to liability claims. For procurement managers, understanding a supplier’s QMS is not just about verifying certification; it is about confirming that quality is proactively managed at every stage.
To make this evaluation actionable, approach supplier discussions with a clear set of questions that cut to the heart of their quality controls: How do they verify the quality and integrity of their raw materials before acceptance? What in-process controls and inspections are performed during manufacturing to maintain consistency? Can they provide documented evidence of rigorous testing protocols on both raw inputs and finished products? How do they maintain and store quality records to ensure traceability over time? Lastly, is there a continuous improvement mechanism that addresses non-conformances and drives corrective actions? These questions help transform a QMS from a theoretical concept into a practical risk mitigation tool, providing you with measurable assurance of supplier reliability.
What is a QMS and Why Does It Matter?
A Quality Management System (QMS) is a formalized framework of documented processes, procedures, and defined responsibilities designed to help an organization achieve its quality policies and objectives consistently. It acts as a blueprint ensuring that every step—from raw material sourcing through manufacturing to final inspection—is governed by standardized practices aimed at maintaining product integrity and meeting regulatory requirements. Rather than relying on reactive problem-solving after defects occur, a robust QMS demonstrates a supplier’s proactive commitment to embedding quality into every phase of production.
One of the most recognized indicators of a mature QMS is certification to the ISO 9001 standard, which sets internationally accepted criteria for quality management systems. Suppliers with ISO 9001 certification have undergone rigorous audits verifying their capability to consistently deliver products that meet customer and regulatory demands. For procurement and compliance officers in the North American hygiene product market, verifying a supplier’s adherence to ISO quality standards is a crucial step in mitigating risk and ensuring dependable product safety and compliance.
Sustainable Hygiene Solutions for Your Business Needs
Top Source Hygiene offers eco-friendly and customizable hygiene products specifically designed for North American markets. From toilet paper to facial tissues, our solutions address sustainability, quality, and cost-effectiveness, making us your trusted partner in the industry.
Key Components to Scrutinize in a Supplier’s QMS
When evaluating a supplier’s Quality Management System (QMS), start by investigating their supplier verification processes. Understand how they select and audit their own raw material suppliers to ensure consistent quality from the very beginning of the supply chain; request documentation of audits, certifications, or performance evaluations that support their vetting process. Next, probe into the process controls they have implemented during manufacturing, such as line inspections, equipment calibration schedules, and production monitoring protocols — these are essential to maintain product consistency and avoid variability that could lead to liability issues.
Further, inquire about their testing protocols, asking for evidence of internal testing conducted on both incoming raw materials and finished goods, including retained sample testing and routine quality checks, supported by documented procedures and test results. Equally important is their record keeping system: determine how quality records, production data, and test results are stored, the duration data is retained, and the ease with which specific batch information can be retrieved — this traceability is vital if a product investigation arises. Lastly, ensure the supplier has a structured continuous improvement process in place, with formal mechanisms for identifying non-conformances, documenting corrective actions, and implementing preventive measures, demonstrating their commitment to evolving and maintaining high quality standards over time.
The Role of Configuration Management: Preventing Unapproved Changes
In simple terms, configuration management is a safety system that prevents unauthorized changes to a product. Think of it as the “master recipe” for your product, where every ingredient and every manufacturing step is documented and locked.
For a hygiene product like toilet paper, this means any potential change—no matter how small—must go through a formal approval process. For example, a supplier cannot unilaterally decide to:
- Switch from 100% virgin wood pulp to a mixed or recycled fiber.
- Change the adhesive used to bind the paper plies.
- Alter the chemical additives that affect softness or strength.
- Modify the packaging material, which could affect product integrity during shipping.
Without this strict change control, a seemingly minor adjustment could lead to widespread issues like skin irritation, reduced product performance, or regulatory violations. For procurement officers, verifying your supplier has a rigorous configuration management process is your assurance that the product you receive in December is identical in quality and safety to the one you approved in January.
The Core of Your Defense: Building a Bulletproof Traceability System
Full batch traceability is the essential operational backbone that transforms what could be a large-scale product liability crisis into a manageable, targeted response. By meticulously documenting every key stage of the product lifecycle—from raw material receipt through manufacturing, packaging, and final shipment—a company gains the ability to quickly and precisely isolate affected batches. This precision prevents costly blanket recalls, limits financial exposure, and safeguards brand reputation in high-stakes markets such as North America.
Traceability begins the moment raw materials arrive at the supplier’s facility, where each lot number and supplier detail is logged. As production proceeds, data such as manufacturing date, production line, shift, process parameters, and packaging identifiers are recorded and linked to the material batch. Finally, distribution details including shipment IDs and destinations complete the chain. This comprehensive documentation enables seamless forward and backward tracking, making it possible to pinpoint exactly which products need to be recalled and which remain unaffected—turning a potential catastrophe into a controlled, verifiable incident.
Infographic: The Journey of a Single Batch
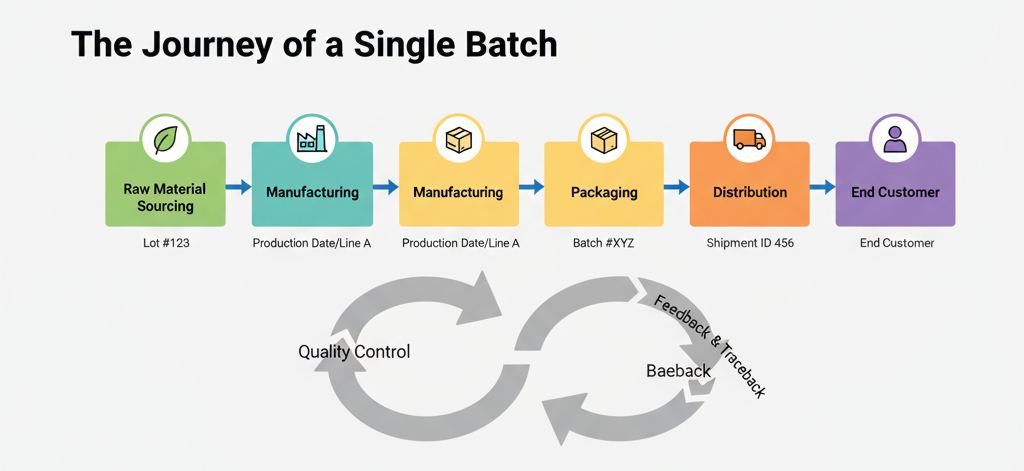
A flowchart tracks a product from Raw Material Sourcing (Lot #123) → Manufacturing (Production Date/Line A) → Packaging (Batch #XYZ) → Distribution (Shipment ID 456) → End Customer.
From Raw Material to Final Delivery: Mapping the Product Lifecycle
Traceability in the hygiene products industry involves systematically documenting every phase of a product’s lifecycle—from the moment raw materials arrive at the factory to the final delivery to customers. This meticulous record-keeping ensures that every item produced can be tracked through its supply chain journey, supporting accountability and quick response in case of any quality issue. By capturing and linking detailed data at each step, companies establish a reliable chain of custody that underpins both internal quality assurance and external compliance requirements.
Key data points critical to effective traceability include the raw material supplier and their respective lot numbers, as these link ingredients or components to their origin. Production details such as the date, shift, and specific manufacturing line used help pinpoint exactly when and where the product was made. Finally, assigning batch or case numbers during the packaging stage completes the traceability record, allowing suppliers and purchasers to trace forward—from raw materials to the end customer—or backward—from a finished product back to its source materials. This linked data network is essential to isolate issues precisely and expediently, minimizing disruption and protecting brand integrity.
How Batch Traceability Minimizes Recall Scope
Before implementing batch traceability, a single defective hygiene product typically forced companies to issue broad recalls covering entire production lots or even all inventory, resulting in significant financial loss and operational disruption. Without the ability to pinpoint the precise batch or raw material origin, manufacturers faced the daunting task of pulling large quantities from the market, which amplified costs and damaged customer trust. After adopting a rigorous batch traceability system, however, companies can quickly isolate problematic products by tracking each item back through raw material lots, production dates, and packaging data. This precise containment not only reduces the recall’s financial impact but also minimizes market disruption and accelerates resolution, thereby preserving brand reputation and customer loyalty.
A well-executed batch traceability system also serves as robust evidence of due diligence to regulators and auditors, demonstrating proactive supply chain control and compliance with safety standards. For example, a North American hygiene product manufacturer recently received an isolated customer complaint regarding skin irritation. Leveraging their batch traceability records, they identified the affected production line and raw material batch within hours, enabling a targeted recall of only 2% of total inventory. This swift action avoided a costly full-lot recall, reassured regulators, maintained retailer confidence, and ensured business continuity. Such operational discipline exemplifies effective supply chain risk management, transforming potential liability into a competitive strength.
Assembling Your Proof: The Technical File and Third-Party Validation
The technical file is your comprehensive dossier that gathers all critical documentation demonstrating a hygiene product’s safety, compliance, and quality control to regulators, auditors, and legal authorities. This file typically includes material safety data sheets (MSDS), detailed manufacturing process flowcharts, batch traceability records, third-party test reports, and relevant certifications such as ISO 9001. Maintaining this file as a dynamic, living document is essential; it must be regularly updated to reflect any product changes, supplier adjustments, or evolving regulatory requirements to ensure ongoing compliance and swift response capability in case of a liability claim.
When reviewing third-party test reports within your technical file, it’s important to go beyond merely confirming their existence. First, verify that the laboratory conducting the tests holds appropriate accreditation and is recognized for impartiality and expertise, such as SGS or Intertek. Next, check the report’s dates to ensure the testing is recent and corresponds to the current production batch or formulation. Additionally, scrutinize whether the samples tested are representative of the bulk product rather than isolated pre-production samples, as this impacts the reliability of the results. Finally, confirm the product was tested against relevant North American standards, and prioritize clear pass/fail outcomes rather than ambiguous or borderline results to confidently uphold your due diligence obligations.
The Anatomy of a Defensible Technical File
A technical file serves as a comprehensive dossier of evidence demonstrating a hygiene product’s safety, compliance, and conformity to applicable regulations and standards. It is an essential component of due diligence in the North American market, where strict liability requires manufacturers and suppliers to substantiate the integrity of their products with clear, organized documentation. This file acts as your primary defense in the event of legal scrutiny, regulatory inquiries, or quality disputes, showcasing a transparent, traceable history of how the product is made and tested.
For hygiene products, a defensible technical file typically includes critical documents such as Material Safety Data Sheets (MSDS/SDS) verifying the safety of raw materials, a detailed Manufacturing Process Flowchart illustrating key quality control checkpoints, and Batch Traceability Records that provide proof of effective tracking throughout production. Additionally, Third-Party Test Reports offer independent validation of product safety and regulatory compliance, while certifications like ISO 9001 evidence a supplier’s formalized quality management system. Importantly, the technical file should be treated as a living document—regularly updated to reflect any product modifications, process changes, or evolving regulatory requirements—to ensure continuous compliance and minimize liability risks.
| Document Type | Purpose & What to Look For |
|---|---|
| Material Safety Data Sheets (MSDS/SDS) | Confirms chemical composition and safety of all raw materials. Check for completeness and recent dates. |
| Manufacturing Process Flowchart | Shows a clear, step-by-step production process. Look for quality control checkpoints. |
| Batch Traceability Records | Proof of a functioning system. Request an example of a mock trace-back report. |
| Third-Party Test Reports | Independent validation of product safety and compliance with specific standards. |
| Certifications (e.g., ISO 9001) | Evidence of a supplier’s commitment to a formal, audited quality management system. |
Leveraging Third-Party Testing for Independent Verification
Independent laboratories such as SGS, Intertek, and TÜV play a crucial role in providing impartial assessments of hygiene products, offering an extra layer of assurance beyond a supplier’s internal quality controls. Their unbiased evaluations are essential because they validate the supplier’s internal claims, helping procurement officers and compliance managers confidently verify product safety and regulatory compliance. By relying on third-party testing, companies can substantiate their due diligence efforts, mitigating liability risks tied to product defects.
Common tests performed on hygiene products include skin sensitivity and dermatological assessments to ensure consumer safety, as well as analyses for restricted substances like heavy metals and formaldehyde that could pose health hazards or regulatory violations. Performance testing further evaluates product effectiveness and durability under typical usage scenarios, confirming that products meet promised specifications. Incorporating these independent test results into your supplier vetting process is indispensable.
How to Critically Review a Test Report
When reviewing a third-party test report, the first critical step is to verify the credibility of the laboratory issuing the report. Ensure the lab is accredited by recognized bodies and has a solid reputation for impartiality and accuracy, as this underpins the reliability of the test outcomes. Next, check the dates carefully to confirm that the report reflects current production batches and relevant testing timelines, since outdated reports might not apply to present manufacturing conditions.
It is equally important to confirm that the product was tested against the appropriate North American standards, which differ from those in other global markets. This ensures the results are directly applicable to your legal and regulatory environment. Finally, scrutinize the test results to verify a clear ‘Pass’ outcome; watch for ambiguous wording or borderline findings that could mask potential issues. A thorough, critical assessment of these elements ensures your technical file genuinely supports your product liability defense and quality assurance efforts.
Tailored Toilet Paper Solutions for Your Brand
Partner with Top Source Hygiene to create premium, customizable toilet paper products backed by 30 years of manufacturing expertise, global delivery, and strict quality standards.

Conclusion: From Risk Mitigation to Competitive Advantage
Navigating product liability in North America requires a proactive, evidence-based approach centered on documentation. The foundation of this approach is a robust supplier QMS that ensures quality is consistently managed. This is complemented by full batch traceability, which provides the precision needed to contain issues, and a well-maintained technical file, supported by third-party testing, to serve as your ultimate proof of due diligence.
By embedding these principles into your procurement process, you move beyond simple risk mitigation. You build a resilient, trustworthy supply chain that becomes a competitive advantage, assuring your customers and stakeholders of your unwavering commitment to safety and quality. Start Your Custom Order →
Frequently Asked Questions
What is a “technical file” and why is it important for the North American market?
A technical file is a meticulously compiled dossier that documents every vital aspect of a hygiene product’s safety, quality, and regulatory compliance. It serves as irrefutable proof that your product meets all necessary standards, providing manufacturers, suppliers, and buyers with transparency and confidence. For companies operating in the North American market, maintaining a thorough technical file is not merely best practice—it is a crucial liability shield that can protect your business in the event of legal scrutiny or product liability claims.
Within the technical file, you’ll typically find essential documents such as Material Safety Data Sheets (MSDS), detailed manufacturing process flowcharts, batch traceability records, and independent third-party test reports. These documents collectively demonstrate due diligence by verifying product composition, outlining controlled production procedures, and confirming compliance with safety standards. In a region governed by strict liability laws, having this organized and accessible evidence can be the difference between efficiently managing a product concern and facing costly recalls, fines, or reputational damage.
How does batch traceability protect my business in case of a product issue?
Batch traceability is a critical safeguard that enables you to pinpoint the exact production details of a hygiene product, including its manufacturing date, production line, and the specific raw material lots used. This detailed tracking system allows you to isolate any affected batch swiftly and accurately in the event of a customer complaint or suspected defect, significantly reducing the scope of products involved in a recall.
By limiting recalls to only the impacted batches instead of an entire inventory, batch traceability minimizes financial losses, helps preserve customer trust, and limits reputational damage. Furthermore, it demonstrates to regulators your proactive control over your supply chain, reinforcing your commitment to quality and compliance in a highly regulated North American market.
What should I look for in a third-party test report?
When reviewing a third-party test report, start by verifying that it originates from a reputable and accredited laboratory such as SGS, Intertek, or TÜV. These labs follow stringent methodologies and impartial protocols, ensuring the credibility of the results. Equally important is confirming that the tests conducted are directly relevant to your specific hygiene product and its intended North American market, covering aspects like chemical composition, skin sensitivity, and compliance with regional regulatory standards.
Carefully examine the test outcomes to ensure the product clearly meets or exceeds the required benchmarks without any ambiguous or borderline results that could undermine your liability defense. A clear “Pass” indication on critical tests shows that the product adheres to safety and quality standards, giving you confidence in your supplier’s claims and fortifying your risk mitigation strategy. Remember, a thorough and well-documented third-party test report not only supports compliance but also serves as vital evidence in demonstrating due diligence if product liability issues arise.


