Dermatological validation sourcing is a non-negotiable requirement for distributors protecting themselves against contact dermatitis liabilities and expensive product recalls. In sectors like mother-baby care and medical procurement, choosing unverified materials often leads to costly insurance disputes and the immediate loss of high-stakes institutional contracts.
Our manufacturing framework uses 100% virgin wood pulp and OBA-free processing to eliminate chemical irritants. We benchmark every batch against Repeat Insult Patch Test (RIPT) data and ISO 9001 standards to ensure a skin-safe pH of 5.5 for sensitive hygiene applications.
Why Do Professional Hygiene Products Require Clinical Validation?
Clinical validation prevents contact dermatitis and liability by ensuring OBA-free materials meet global safety benchmarks for prolonged skin contact in professional environments.
Preventing Adverse Skin Reactions in High-Traffic Environments
Professional environments like hotels and hospitals require verified skin safety to prevent contact dermatitis. Clinical testing identifies allergens and ensures fiber structures do not cause micro-abrasions during frequent use.
- RIPT Testing: We use the Repeat Insult Patch Test (RIPT) to identify allergens that cause Type IV allergic contact dermatitis in sensitive users.
- Fiber Integrity: Dermatological assessments verify that the paper texture remains smooth, preventing micro-abrasions during high-frequency use.
- 2026 Standards: Aligning with upcoming safety benchmarks reduces legal risks and insurance liability for institutional buyers.
- Sector Specifics: Hospitality and healthcare settings demand non-irritating structures due to the volume of daily skin contact.
Certifying Safety via OBA-Free Virgin Wood Pulp Standards
Sourcing 100% Virgin Wood Pulp from our Hebei industrial hub provides a natural baseline that avoids chemical risks. We eliminate secondary additives to ensure compatibility with compromised skin barriers.
- Material Purity: 100% Virgin Wood Pulp provides a naturally lint-free experience without the contaminants found in recycled waste.
- OBA-Free Manufacturing: We guarantee the absence of Optical Brightening Agents to prevent chemical leaching and skin sensitization.
- Regulatory Compliance: All processes meet ISO 9001, FDA, and EU hygiene standards for a verified safety profile.
- Environmental Validation: Rapid disintegration testing ensures the product remains septic-safe while meeting international plumbing standards.
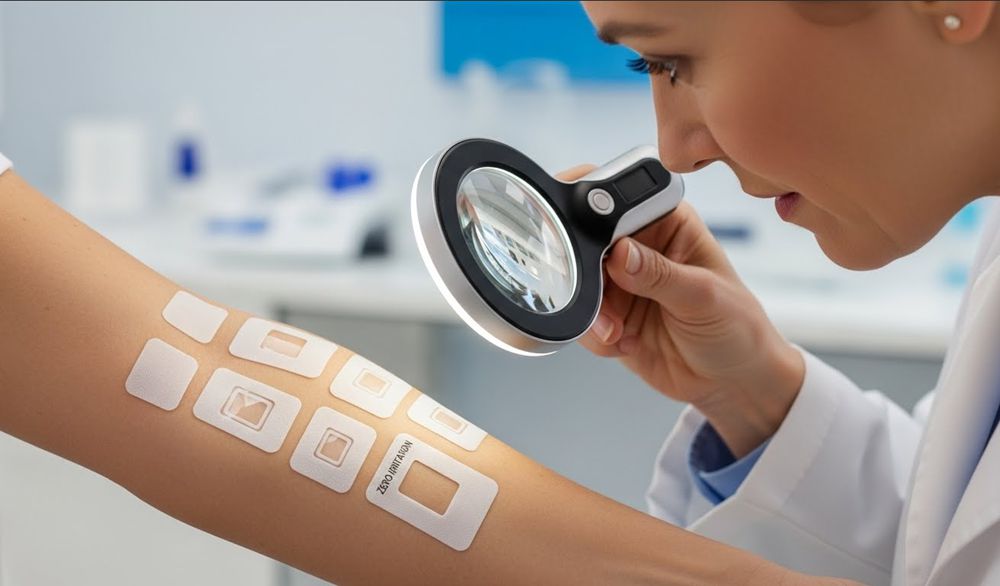
What is an RIPT and How Does it Test for Skin Sensitization?
A 6-week clinical study identifying potential allergens through induction, rest, and challenge phases to validate the dermatological safety of virgin wood pulp hygiene products.
| RIPT Phase | Duration | Clinical Objective |
|---|---|---|
| Induction Phase | 3 Weeks | Repeated exposure to promote material penetration. |
| Rest Period | 21 Days | Allowing the immune system to develop latent sensitization. |
| Challenge Phase | 48–72 Hours | Testing naive skin sites to detect allergic contact dermatitis. |
The Three Phases of Clinical Sensitization Testing
The Repeat Insult Patch Test (RIPT) serves as the industry benchmark for verifying that topical materials do not trigger Type IV allergic reactions. Clinicians execute this study over six weeks to monitor how human skin reacts to repeated exposure under controlled, occlusive conditions.
- Induction Phase: Clinicians apply ten repeated patches over 48-72 hours each to the same site to encourage potential sensitization.
- Rest Period: Subjects undergo a 21-day hiatus where no product is applied, giving the immune system time to recognize any potential allergens.
- Challenge Phase: The lab applies two consecutive 48-hour patches to previously untested skin sites to confirm if an allergic response exists.
- Grading System: Experts evaluate reactions using a 5-point scale, ranging from 0 (no sign of irritation) to 4 (erythema with edema and blistering).
Safety Validation for OBA-Free Virgin Wood Pulp
For B2B buyers in the hospitality and medical sectors, RIPT data confirms that paper materials meet strict hypoallergenic standards. We focus on high-purity fibers that maintain skin barrier integrity without introducing chemical irritants commonly found in lower-grade recycled alternatives.
- 100% Virgin Wood Pulp: Clinical testing verifies that long-fiber wood pulp remains non-irritating even under the high-pressure conditions of an occlusive patch.
- OBA-Free Guarantee: We eliminate Optical Brightening Agents to prevent Type IV contact dermatitis triggered by fluorescent whiteners.
- Ply Density Control: Maintaining a 13–22g/m² ply density ensures structural integrity, reducing mechanical friction that can cause redness in sensitive skin.
- Institutional Compliance: RIPT results provide the necessary evidence for hypoallergenic claims required by global retail chains and luxury hotel groups.
Premium Custom Toilet Paper at Wholesale Prices

How to Evaluate PH Neutrality (Target 5.5) for Sensitive Care?
Validating pH 5.5 protects the skin’s acid mantle. Precision testing and using 100% OBA-free virgin wood pulp ensure product safety for sensitive hygiene applications.
Testing Protocols for Skin-Compatible Acidity
Professional hygiene validation moves beyond basic litmus paper. Real-world accuracy requires digital equipment and standardized preparation to ensure every production batch aligns with the skin’s natural acidic mantle. This slightly acidic environment prevents bacterial colonization and maintains the moisture barrier.
- Digital Meter Calibration: Use standard solutions to calibrate meters for an accuracy range between 5.4 and 5.9.
- 10% Dilution Method: Mix 10g of paper material with 90mL of distilled water to create a stable solution for testing solid products.
- Therapeutic Monitoring: Maintain pH levels above 5.0 to support re-epithelialization in compromised skin and prevent pathogenic growth like Staphylococcus aureus.
Ensuring Microbiome Safety with OBA-Free Virgin Pulp
Material purity is the foundation of pH stability. We eliminate secondary chemical treatments that typically fluctuate acidity or cause sensitization. For B2B distributors, providing evidence of fiber purity is often more persuasive than a simple “dermatologically tested” label.
- 100% OBA-Free Material: We exclude Optical Brightening Agents and fluorescent whiteners to prevent chemical contact dermatitis.
- Natural Brightness: Our 100% virgin wood pulp achieves 85-92% brightness through fiber selection rather than aggressive chemical bleaching.
- Regulatory Alignment: Manufacturing processes meet ISO 9001, FDA, and EU hygiene standards to ensure compatibility with sensitive microbiome environments.
- Septic Safety: High-purity fibers are engineered for rapid disintegration, meeting North American and European plumbing standards.
What are the ISO 10993 Standards for Cytotoxicity Testing?
ISO 10993-5 evaluates material-cell interactions. It classifies substances as cytotoxic if they reduce cell viability by over 30% or exceed a numerical reactivity grade of 2.
Cell Viability Thresholds and Quantitative Grading
ISO 10993-5 provides a standardized framework to determine if a material damages human cells upon contact. This screening is essential for hygiene products that touch sensitive skin areas for extended periods.
- 0–4 Grading Scale: Grade 0 represents zero reactivity; Grade 4 indicates severe, nearly complete destruction of the cell layers.
- 30% Failure Limit: Quantitative assays classify any material that reduces cell viability by more than 30% as a cytotoxic failure.
- Testing Protocols: Laboratories use direct contact, agar diffusion, or liquid extraction to measure how mammalian cells respond to the material.
- Detection Sensitivity: These protocols identify toxic residues and chemical leachates even when they exist in very low concentrations.
OBA-Free Virgin Wood Pulp and Biocompatibility
Material purity directly influences cytotoxicity results. At Top Source Hygiene, we align our manufacturing with these international safety benchmarks to protect end-user skin health and distributor reputation.
- 100% Virgin Wood Pulp: Using primary fibers removes the risks associated with recycled pulp, which often contains ink residues or adhesives that trigger cytotoxic reactions.
- OBA-Free Production: We eliminate Optical Brightening Agents and fluorescent whiteners. This prevents chemical interference with the skin microbiome.
- Regulatory Compliance: Strict adherence to ISO 9001 and FDA standards validates the safety of our 5-ply ultra-soft rolls for the luxury hospitality sector.
- Natural Brightness: We achieve 85-92% brightness through careful fiber selection, avoiding the need for aggressive, cytotoxic chemical bleaching agents.

How to Market “Dermatologist-Approved” Status to B2B Clients?
B2B buyers view clinical validation as a risk-mitigation tool. Focus on material purity and OBA-free standards to secure high-stakes contracts with healthcare providers and premium retail groups.
Strengthening B2B Distribution Through Clinical Validation Badges
B2B buyers in the pharmacy and medical sectors rarely care about “softness” alone; they prioritize clinical certainty. Professional procurement officers view dermatologist endorsements as an insurance policy against product liability and customer complaints. When pitching to specialty retail stores, lead with the fact that these badges represent a passed Repeat Insult Patch Test (RIPT), the industry gold standard for assessing skin sensitization.
- Institutional Procurement: Feature dermatologist-tested ratings on all B2B documentation to provide immediate scientific credibility for hospital and clinic buyers.
- Pharmacy Distribution: Position endorsements as a mandatory competitive advantage when bidding for shelf space in medical-grade retail environments.
- Targeted Advertising: Use clinical validation data in professional campaigns aimed at dermatology practices and institutional procurement officers.
- Bulk Reassurance: Utilize social proof and clinical ratings in email marketing to reduce the perceived risk for wholesalers evaluating products for long-term resale.
Strategic Positioning of OBA-Free and 100% Virgin Wood Pulp Standards
We align “dermatologist-approved” status with the material integrity of our Top Source Hygiene production lines. Marketing unmedicated tissue products to business clients requires translating clinical safety into technical specifications. This means proving that the paper itself—rather than added chemicals—provides the safety profile required for sensitive skin.
- 100% OBA-Free Guarantee: Market the absence of Optical Brightening Agents to eliminate the risk of chemical irritation in sensitive clinical environments.
- Material Purity: Emphasize high-purity, long-fiber virgin wood pulp as a lint-free alternative to recycled materials, which often contain hidden chemical residues or PFAS.
- Natural Brightness (85-92%): Highlight that visual quality comes from superior fiber selection rather than aggressive chemical bleaching processes.
- Clinical Compliance: Align our rapid disintegration and septic-safe technology with the rigorous hygiene and maintenance requirements of high-traffic healthcare facilities.
- pH Compatibility: Connect the product’s natural profile to the skin’s acidic pH 5.5 requirement, supporting the acid mantle in long-term care settings.
Frequently Asked Questions
What does a ‘Dermatologically Tested’ claim signify for B2B paper sourcing?
This label indicates that the product was evaluated on human subjects under the supervision of a dermatologist, typically involving 30 participants over a 72-hour monitoring period. Since there are no universal regulatory standards for this claim, professional buyers should request specific laboratory protocols and third-party certifications, such as those from Dermatest®, to verify the depth of the assessment.
What is the clinical role of a Repeat Insult Patch Test (RIPT) in tissue manufacturing?
RIPT is a standardized clinical procedure used to identify potential skin sensitization and allergic contact dermatitis. The test involves an induction phase with repeated applications over three weeks, followed by a rest period and a final challenge phase. This rigorous process allows manufacturers like Top Source Hygiene to ensure that high-ply virgin wood pulp products are safe for prolonged skin contact before entering mass distribution.
Why is a slightly acidic pH of 5.5 preferred over a neutral pH of 7.0 for hygiene products?
While pH 7.0 is chemically neutral, healthy human skin maintains an ‘acid mantle’ with a pH around 4.7 to 5.5. Products balanced to pH 5.5 support this natural barrier, aiding in antimicrobial defense and moisture retention. Using neutral or alkaline products can disrupt this mantle, leading to dryness, inflammation, and increased susceptibility to bacterial growth.
Does ‘unscented’ mean a toilet paper product is hypoallergenic?
No. ‘Unscented’ products often use masking chemicals to neutralize odors, which can still trigger irritation. For users with sensitive skin, ‘fragrance-free’ is the safer standard as it indicates no perfumes or masking agents were added. For 2026 procurement standards, distributors should also prioritize OBA-free (no optical brightening agents) and chlorine-free certifications to ensure true hypoallergenic performance.
Which chemical contaminants are most common in unverified, low-cost tissue products?
Research indicates that nearly 46% of certain budget tissue brands contain detectable levels of PFAS (forever chemicals), often used for moisture resistance. Other risks include trace formaldehyde used for wet-strength and dioxins from elemental chlorine bleaching. High-end manufacturers mitigate these risks by using 100% virgin wood pulp and oxygen-based whitening processes.
How can distributors leverage dermatological safety data to secure medical or high-end retail bids?
Success in institutional bidding requires presenting clinical evidence such as HRIPT results and ISO 10993 cytotoxicity data. Using standardized safety reporting, like the Medical Dictionary for Regulatory Activities (MedDRA), and aligning data with American Academy of Dermatology (AAD) standards demonstrates a commitment to transparency that satisfies the risk-benefit requirements of procurement committees.


